Soft Tissue Restoration, Blood Vessel Formation Focus of $3M Grant
07/11/2023
UNIVERSITY PARK, Pa. — The ability to regenerate and pattern blood vessels, the literal lifelines extending deep into soft tissues, remains an elusive milestone in regenerative medicine. Known as tissue revascularization, stimulating blood vessel growth and pattern formation in damaged or diseased tissues could accelerate the field of regenerative medicine, according to Penn State researchers.
With a four-year, $3 million grant awarded by the National Institutes of Health’s National Heart, Lung, and Blood Institute, Penn State chemical engineering and reconstructive surgery researchers plan to develop a new way to help restore soft tissue loss in patients through two coordinating revascularization techniques.
“Tissue revascularization is a bottleneck for regenerative medicine,” said principal investigator Amir Sheikhi, assistant professor of chemical engineering in the College of Engineering, who also has an affiliation with biomedical engineering. “This is an important award for the whole field, as we hope to develop a fundamentally new way to tackle the problem using a transdisciplinary team.”
When repairing a traumatic injury, surgeons must be able to restore blood flow rapidly to grafts, flaps and engineered scaffolds. However, this is not always feasible using conventional techniques, according to researchers.
The researchers plan to combine a class of protein-based granular hydrogel biomaterials pioneered by Sheikhi, with a microsurgical tactic known as vascular micropuncture, developed by co-principal investigator Dino Ravnic, Huck Chair in Regenerative Medicine and Surgical Sciences, associate professor of surgery at the Penn State College of Medicine, and an attending plastic surgeon at the Penn State Health Milton S. Hershey Medical Center.
Bulk hydrogel scaffolds — polymer networks that can hold a large amount of water while maintaining their structure — have been used over the past few decades as a platform to restore soft tissues during surgical repair, according to Sheikhi, but they often suffer slow and random vascularization effects upon implantation.
To address the limitations of bulk hydrogels, Sheikhi said he plans to engineer protein-based granular hydrogel scaffolds by attaching microscale hydrogel particles to each other.
“By adjusting the empty spaces among the hydrogel particles, we can regulate how cells interact with each other and assemble, guiding tissue architecture and the formation of new blood vessels,” Sheikhi said.
At the same time, researchers will implement vascular micropuncture, where Ravnic and his team will puncture blood vessels with microneedles to accelerate the formation of new blood vessels. The tiny size of the needles ensures there is no blood clotting or significant bleeding.
“Our microsurgical approach allows for targeted blood vessel formation without the use of any added growth factors or molecules,” Ravnic said. “This is exceedingly relevant to advancing tissue engineering and also in treating blood vessel-related conditions.”
The researchers will first test their approach using human cells cultured in vitro from patient samples. Once they establish a baseline understanding of the approach at the cellular level, they will test it in rodents.
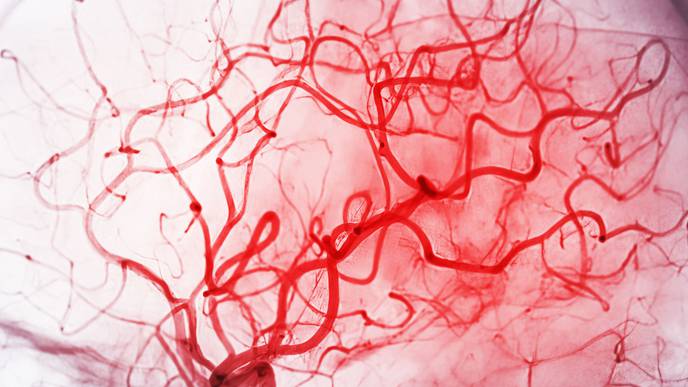
The combination of the two techniques, researchers predict, will allow for new blood vessels to rapidly form in an architecturally organized manner. The hierarchical formation — the organization of blood vessels from big to medium to small — helps regulate blood flow, diffuse oxygen and modulate immune cells throughout reconstructed or injured soft tissue.
“The patterns of blood vessels should resemble tree branches, with a large trunk fanning out into smaller and smaller branches,” Sheikhi said. “The reason is that blood needs to flow from the main vessels deep within tissues through capillaries.”
Shayn Peirce-Cottler, professor and chair of biomedical engineering at the University of Virginia, will collaborate on the grant.

Facebook Comments